
How Cxbladder works
Research from Pacific Edge shows that levels of specific messenger RNA (mRNA) and DNA biomarkers are present at different levels in urine samples from patients who have urothelial cancer than in patients who don’t.
Cxbladder tests harness this research by measuring the gene expression of the biomarkers present in a sample of the patient's urine. The relative levels of gene expression allows us to predict a person's likelihood of having urothelial cancer.
What biomarkers does Cxbladder analyze?
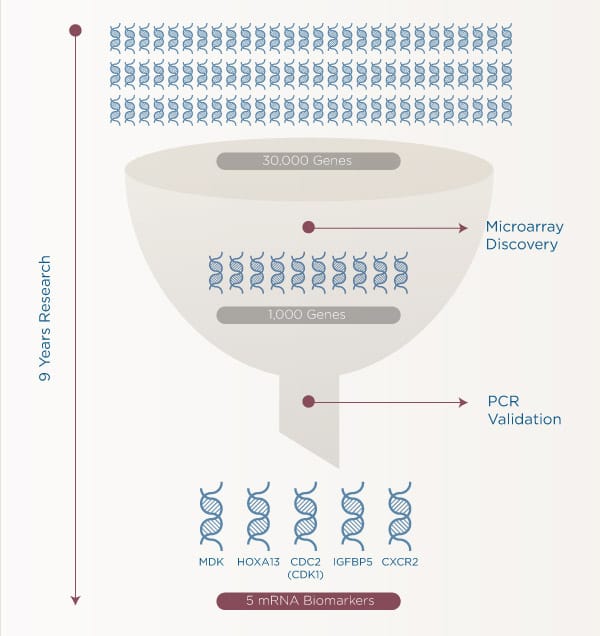
Cxbladder tests analyze five mRNA signatures in a patient's urine:
MDK: Blood vessel growth and cell migration. Principally involved in cell proliferation, migration and angiogenesis in cancer cells.
HOXA13: Cell differentiation. Principally involved in cell differentiation and the morphogenesis and differentiation of the genitourinary tracts.
CDC2 (CDK1): Cell division. Cyclin dependent kinase. Essential to mitotic cell cycle: cell proliferation
IGFBP5: Programmed cell death. Acts as an anti-apoptotic gene.
CXCR2: Inflammation. Mediates neutrophil migration to sites of inflammation. Moderates non-malignant inflammation (reduces False Positives).
Cxbladder Triage Plus, a next generation Cxbladder test, additionally analyzes 6 DNA single nucleotide polymorphisms (SNPs) from 2 genes to enhance performance:
FGFR3: Cell proliferation, differentiation and apoptosis
TERT: Telomere elongation, cellular senescence
How does testing work?
A small sample (5 ml) of mid-stream urine is required for the test. Once collected using our provided system and sent to our laboratory, the sample undergoes a precise set of processes to extract and purify mRNA and DNA present.
In the Cxbladder test, each biomarker of interest is quantified by a different probe, and the relationship between the individual biomarkers is determined with a mathematical equation. The calculated outcome provides a measure of the probability of the presence of urothelial carcinoma.
What makes Cxbladder so accurate?
Cxbladder calculates probability objectively, without relying on people to count cells manually, so the results of this urine gene expression test are reliable.
With high Negative Predictive Value (NPV) and high Sensitivity, tests in the Cxbladder suite are optimized for the risk stratification of urothelial cancer, and UC rule out in patients with a low risk of disease.
Cxbladder is a precision test that delivers exceptional performance and clinically actionable results
The Cxbladder suite is supported by an evidence portfolio that includes over 30 peer reviewed publications in leading medical journals.
Cxbladder Triage is now included in the AUA/SUFU Microhematuria Guideline, recommended with supporting "Grade A" Evidence*.
The Cxbladder suite of tests have been used by over 5,000 urologists in more than 130,000 patients.**
Start your Cxbladder journey
Contact our customer service team
Learn more about the Cxbladder suite and how to implement it in your practice.

The sampling process
Cxbladder’s propriety urine sampling system has been developed with urological staff to ensure ease of use for both patients and clinical teams.

The test report
The Cxbladder test report provides actionable results that are intended to aid in the risk stratification of urothelial cancer when used in conjunction with standard clinical assessment.
The report have been developed in consultation with leading urologists and provides a patient’s validated Cxbladder score, alongside an interpretation of the results and an assay description.

Optimize your clinical practice while improving the patient experience
The Cxbladder suite helps you prioritize your time and clinical resources on those that need it the most, streamlining practice workflow and increasing overall efficiency.
Cxbladder works for both you and the patient. Spare patients with a low probability of urothelial cancer the discomfort, anxiety and risks of an invasive procedure, while optimizing your clinical practice while delivering the best patient experience possible.
Adopting Cxbladder in your clinic

Implementing Cxbladder
Our team are ready to guide and support you in the deployment and use of Cxbladder. Learn more implementation.

Start your Cxbladder journey
Contact our customer service team to learn more about the Cxbladder suite and how to implement it in your practice.

Cxbladder is available around the world
Cxbladder is available in the United States, and in countries across Asia and South America. Cxbladder is also available in New Zealand, Australia, and Israel.
References
* Barocas DA, Lotan Y, Matulewicz RS, Raman JD, Westerman ME, Kirkby E, Pak L, Souter L. Updates to Microhematuria: AUA/SUFU Guideline (2025). J Urol. 0(0). doi: 10.1097/JU.0000000000004490.\
** Pacific Edge data, March 2026

